Aberrant constitutively active NOTCH1 upregulates anabolic pathways and oncogene MYC expression, which promote T cell leukemogenesis. 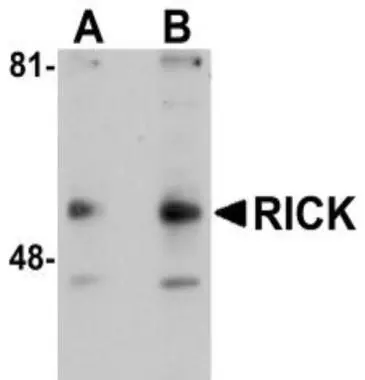
The activation of the NOTCH1 pathway is essential for early T cell fate determination in the haematopoietic system. NOTCH1 activation is triggered by interaction with its ligands, which are members of the Delta and Jagged families and are expressed on neighboring cell surfaces. This clinical challenge has led researchers to decipher the molecular mechanism of T-ALL transformation and progression and develop alternative drugs to treat this malignancy.Ī genome-wide association study of T-ALL cases has identified considerable gene mutations among them the key oncogenic regulator is NOTCH1, which is found in more than 60% of T-ALL. Although the current cure rates increased to 80% in children and 60% in adults, patients with primary resistant T-ALL frequently fail to obtain a complete hematological remission or relapse after the initial response. T-ALL originates from genomically altered and/or epigenetically changed transformation of immature T cells. Analysis time is also reduced.T-cell acute lymphoblastic leukemia (T-ALL) is an aggressive hematological neoplasm that frequently occurs in children and adolescents worldwide. It is especially useful when the target protein band overlaps with the heavy or light chain band. List of tagged protein purification gel and elution peptide setsįor analysis of immunoprecipitated proteinsĬo-eluted antibody is not detected with HRP-labeled antibodies in Western blotting of immunoprecipitated proteins. List of anti-tag antibody-coated magnetic agarose beads List of anti-tag antibody-coated magnetic beads List of anti-tag antibody-coated agarose beads List of antibody-coated magnetic agarose beads Primary antibodies suitable for IP (excluding anti-tag antibodies) >The principle and method of polyacrylamide gel electrophoresis (SDS-PAGE) >The principle and method of Western blotting (WB) Pull-down assay experiments using Tagged protein purification kits ※The antibodies are co-eluted, which should be taken into account when analyzing the data.Įlute the target protein in neutral pH using an elution peptide and avoid harsh conditions, such as acidic and alkaline solutions. Centrifuge, and use the supernatant for SDS-PAGE. 
Repeat 3-4 times.Īdd 50 µL of 2x SDS sample buffer containing 2-mercaptoethanol (2-ME) and heat for 5 minutes to elute the target protein from the beads. Remove the supernatant by aspiration without disturbing the beads. Pellet the agarose beads by centrifugation, and remove the supernatant by aspiration without disturbing the beads.Īdd 1 mL of ice-cold lysis buffer or washing buffer, mix, and centrifuge. Incubate at 4☌ for 1 hour-overnight on a rotating shaker. Incubate at 4☌ for 1 hour-overnight with shaking on a rotator.Īdd secondary antibody-coated (or protein A/G-coated) agarose beads. IP (with agarose beads) ※An example performed at MBLĪdd 500 µL of protein extract and 2-10 µg of the primary antibody to a 1.5-mL tube. Stable oligomeric complexes are formed because reaction between a polyclonal antibody and an antigen is multivalent. If the binding affinity of an antibody is low, simultaneously using several high-specificity monoclonal antibodies will allow multivalent binding, resulting in stable antigen-antibody complexes. 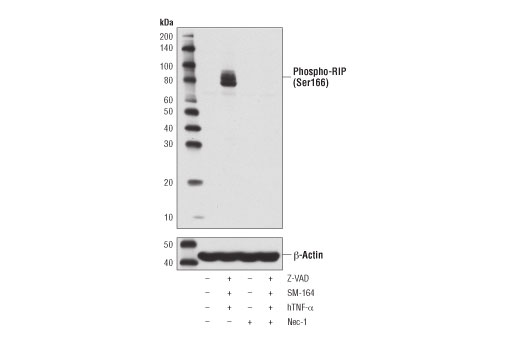
High-affinity monoclonal antibody (dissociation constant K d<10 -8 M) should be used because low affinity antibody may not form an antigen-antibody complex in solution.Įven if the affinity of individual antibody molecules is low, oligomeric antigen-antibody complexes are formed easily due to the multivalent binding. Background is low with an appropriate antibody because the antibody recognizes a single antigen.īackground may be high if some of the antibody molecules have a low specificity and bind to proteins other than the target protein.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
